 Hydrogen Gas Detection Solutions
Hydrogen Gas Detection Solutions
As well as being an important industrial gas, hydrogen is becoming increasingly important as a fuel. However, hydrogen is fundamentally unlike any other fuel source, both in terms of its function and its hazards.1
With the global hydrogen economy continuing to grow, hydrogen is set to play a major role in the decarbonization of the world’s energy supply. But adopting hydrogen technologies requires that robust safety measures are put in place, including the deployment of reliable hydrogen gas detection systems.
The Importance of Hydrogen
Virtually all of the fuels currently used worldwide are derived from naturally occurring oil or gas. These fuels are a finite resource, and their use is known to be harmful to the environment. As a result, the world is shifting toward renewable energy sources.
Increased renewable energy production presents its own challenges: How can energy produced by intermittent sources such as wind and solar be stored? And how can transport and industrial sectors that rely on energy-dense chemical fuels continue to operate in a post-carbon economy?
Hydrogen is arguably the best answer to both of these questions. Today, hydrogen is largely produced by steam methane reforming (SMR) and is widely used as an industrial gas. However, renewable energy can be used to create hydrogen gas via processes such as water electrolysis, effectively locking up that energy within hydrogen molecules. In this capacity, hydrogen acts as an energy storage medium rather than a conventional fuel.
Hydrogen offers numerous advantages over other energy storage technologies. Like conventional fuels, hydrogen has a very high energy density – much higher than batteries or capacitors – and can store energy indefinitely without losses. However, unlike conventional fuels, hydrogen does not produce dangerous emissions at the point of use: the only by-product of burning hydrogen is water. Hydrogen technology is still in its infancy – but there is already reason to believe that hydrogen could become significantly cheaper per unit of energy stored than batteries.1–3
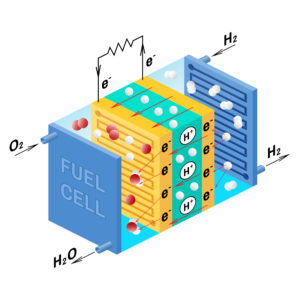
Fuel cell diagram. Vector. Device that converts chemical potential energy into electrical energy. A PEM, Proton Exchange Membrane cell uses hydrogen gas and oxygen gas as fuel.
The result is that the hydrogen economy is growing rapidly. The global hydrogen market is forecast to increase by roughly 6% per year, growing to $155 billion by 2022.4
The Risks of Hydrogen
Increased uptake of hydrogen technologies is not without its downsides. While hydrogen performs well as a fuel, it presents a much greater explosion hazard than many other liquid and gas fuels.
This is for two reasons. Firstly, hydrogen is much more difficult to contain than other gases. Hydrogen gas consists of H2 molecules: each molecule is made up of two hydrogen atoms bound together. This makes H2 the smallest molecule in the universe. Hydrogen gas is therefore prone to leaking out of containment.5
In addition to this, hydrogen is extremely flammable. Hydrogen in air is flammable at concentrations between 4% and 75% by volume (for comparison, methane is flammable in air only in a proportion between 4.4% and 17% by volume). The amount of energy required to ignite a hydrogen/air mixture is also much lower than for other fuels. The minimum amount of energy required to ignite a mixture of hydrogen and air is just 0.017 mJ. In contrast, the minimum ignition energy for hydrocarbon fuel gases is much higher, at around 0.3 mJ for methane/air or propane/air mixtures.6,7
The result is that hydrogen leaks are common, and even very small hydrogen leaks can be relatively easily ignited.8 It is therefore essential that appropriate measures are put in place to mitigate the risk of hydrogen leaks.
Hydrogen Gas Detection Technology
Hydrogen is an odourless, colourless, and tasteless gas. Industry, therefore, relies on hydrogen gas detectors to detect leaks. IGD has two technologies suitable for detecting hydrogen: pellistor sensors and electrochemical sensors.
Pellistor
Pellistor, or catalytic bead, sensors rely on the use of a catalyst that causes flammable gas within the sensor to ignite at a much lower temperature than usual. When combustion occurs, heat is produced in proportion to the amount of flammable gas present. The concentration of flammable gases can then be derived from this measurement and expressed as a percentage of the lower explosive limit (%LEL).

Pictured: TOC-750 safe area addressable gas detector
Pellistor sensors are typically used as a general “catch-all” technology for flammable gas detection. Pellistors respond to any flammable gas, measuring 0-100% LEL. Since a 4% concentration of hydrogen is explosive, this corresponds to 100% LEL. Most legislation (such as the UK Dangerous Substance Explosive Atmosphere Regulations (DSEAR)) requires an atmosphere to be maintained below 25% LEL.
IGD pellistor gas detectors are the world’s most reliable, cost effective and robust of their kind in the market, making them ideal for hydrogen gas detection.
Electrochemical
Electrochemical sensors work by reacting the target gas – in this case, hydrogen – with an electrolyte, which produces a current in proportion to the amount of gas present. This allows for much more sensitive hydrogen gas detection compared to pellistor sensors. For example, 25% LEL equates to 1% hydrogen concentration, or 10,000 ppm. IGD electrochemical gas detectors offer sensitivity in the ranges of 0-1000 ppm to 0-40,000ppm. However, the downside of this extreme sensitivity is that electrochemical sensors can be destroyed on exposure to levels exceeding their measurement range, requiring them to be replaced. IGD electrochemical hydrogen detectors are ideally suited to applications where detection of hydrogen at low levels is critical.
Other Detection Technologies
Several other gas detection technologies exist; however, these are not recommended for hydrogen detection.
Infrared sensors are unable to detect hydrogen since diatomic molecules like hydrogen don’t absorb infrared radiation.
Semiconductor gas detectors can be used to detect hydrogen; however, these sensors also typically respond to a wide range of other gases and vapours. The likelihood of false alarms means that semiconductor sensors are not advised for these applications.
Thermal conductivity is another viable technology, though low sensitivity and selectivity render them poor for hydrogen detection applications.
International Gas Detectors Hydrogen Gas Detection Solutions
International Gas Detectors can provide a range of fixed electrochemical and/or pellistor hydrogen gas detection systems, designed to suit all applications. These include our IP54 addressable 750 safe area detectors used for non-ATEX applications. Our IP68 750X ATEX/IECEx explosion proof detectors. In addition, we can provide TOC-750S addressable aspirated detectors for cleanrooms or hard to access areas. This enables clients to have a true digital gas detection system which is Internet of Things enabled. Packed full of features, benefits and cost savings not seen on any other hydrogen gas detection system available on the market. See our hydrogen gas detection products here.

To complete our fixed hydrogen gas detection offering, we also have our TOC-903-X5 ATEX/IECEx standalone non-intrusive gas detectors. The 903-X5 offers a huge array of benefits, including dual gas sensing capabilities, 5-Port housing and extreme versatility. Find out more about the X5 here.
Portable Hydrogen Gas Detectors
Finally, we provide portable hydrogen monitors for added personal safety and leak identification. These can either be as a single gas monitor at 0-1000ppm to help identify leaks in equipment. Alternatively we also have a standard 4 gas monitor for confined space works.
Contact Us
To find out more about the range of hydrogen gas detectors from IGD – as well as accessories such as gas collector cones & hoods and remote gassing ports – get in touch with a member of the IGD team today.
References and Further Reading
- The Future of Hydrogen – Analysis. IEA https://www.iea.org/reports/the-future-of-hydrogen.
- Pellow, M. A., Emmott, C. J. M., Barnhart, C. J. & Benson, S. M. Hydrogen or batteries for grid storage? A net energy analysis. Energy Environ. Sci. 8, 1938–1952 (2015).
- Batteries and hydrogen technology: keys for a clean energy future – Analysis. IEA https://www.iea.org/articles/batteries-and-hydrogen-technology-keys-for-a-clean-energy-future.
- Bezdek, R. H. The hydrogen economy and jobs of the future. Renew. Energy Environ. Sustain. 4, 1 (2019).
- Lanz, W. Hydrogen Properties. Hydrogen Fuel 47 (2001).
- ISO – ISO/TR 15916:2015 – Basic considerations for the safety of hydrogen systems. https://www.iso.org/standard/56546.html.
- Haase, H. Electrostatic hazards. Their evaluation and control. (Verlag Chemie, 1977).
- Butler, M. S., Moran, C. W., Sunderland, P. B. & Axelbaum, R. L. Limits for hydrogen leaks that can support stable flames. International Journal of Hydrogen Energy 34, 5174–5182 (2009).

 Hydrogen Gas Detection Solutions
Hydrogen Gas Detection Solutions Portable Hydrogen Gas Detector – Buy Online
Portable Hydrogen Gas Detector – Buy Online TOC-750 Addressable Hydrogen Gas Detector
TOC-750 Addressable Hydrogen Gas Detector TOC-750X Addressable Hydrogen Gas Detector
TOC-750X Addressable Hydrogen Gas Detector TOCSIN 903 Standalone ATEX/IECEx detector
TOCSIN 903 Standalone ATEX/IECEx detector


